Microbiome and chronic disorders
The incidence of immune-mediated (IBD, T1D) and/or metabolically-driven (Obesity, T2D) chronic disorders are dramatically increasing over the past decades in industrialized countries. Nutrition-related factors together with the gut microbiome emerged as prime environmental factors in the disease etiology. The search for disease susceptibility genes in the human genome is currently complemented with the analysis of the gut microbiome. Since the pioneering work by Louis Pasteur and Robert Koch in the 19th century, the study of pathogens has been a major interest in the field of microbiology. Yet, over the last twenty years, advances in gut microbial ecology have highlighted the fundamental role of commensal gut microorganisms in health and disease. The microbiome describes the symbiotic ecological community of commensal and pathogenic microorganisms that literally share the body space (Lederberg and McKay 2001 Scientist).
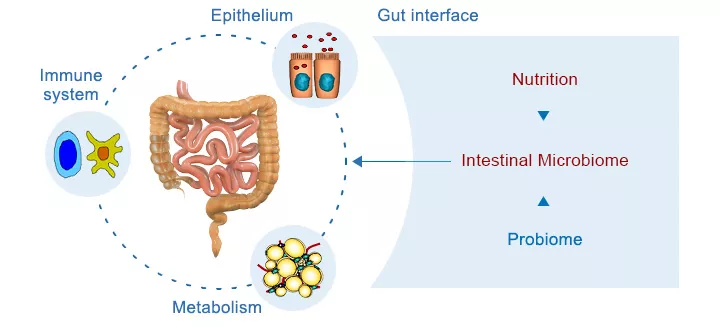
The gut microbiome has a complex reciprocal relationship with its host. Its composition and activity can have both beneficial and deleterious effects, and at the same time, the (patho-)physiological state and genomic make-up of the host can modulate the profile and impact of the microbiome. A detailed description of the microbiome will require data on the genomic profiles, phylogenetic relationships, and spatial and temporal distribution of the different microorganisms that it comprises. The study of intestinal metagenomes by analyzing 16S rRNA gene or complete genome sequences has delivered first descriptive insights into bacterial functions in health and diseases and has shown that gut microbial ecosystems are even more complex than previously assumed. Gnotobiology, i.e., the use of germfree animals, has been essential for the discovery of bacterial functions in the gut. A key focus for research into cellular mechanisms is the tissue-environment interface. In the context of host-microbe interactions at the gut interface, intestinal epithelial cells (IEC) must adapt to constant changes in their environment by processing both bacterial and host-derived immune signals.
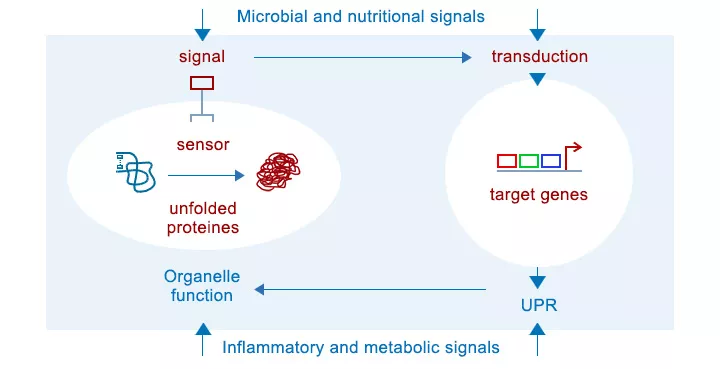
Cell stress und unfolded protein response (UPR) – sensing the environment
An emerging paradigm suggests that cell stress initiated through dysfunctions in the endoplasmatic reticulum (ER) and mitochondria may contribute to the loss of tissue homeostasis and the development of chronic pathologies. Extracellular signals and cellular demands are integrated in the cellular stress responses program orchestered by the unfolded protein response (UPR) of the endoplasmic reticulum (ER-UPR) and mitochondria (Mt-UPR). Protein folding and the maintenance of energy homeostasis are important cellular demands. Failure to adapt to these processes lead to uncontrolled inflammation, hyperproliferation or apoptosis associated with the loss of organ function. It seems intriguing that UPR signaling is essential for tissue homeostasis, but at the same time, cellular stress response mechanisms are implemented in the pathogenesis of IBD, colorectal cancer and diabetes, suggesting that common factors are involved in the development of immune-mediated and/or metabolically driven chronic disorders.